Martin Kaplan, DMD, and Peter Vitruk, PhD, explore soft tissue CO2 laser uses in orthodontics
Introduction
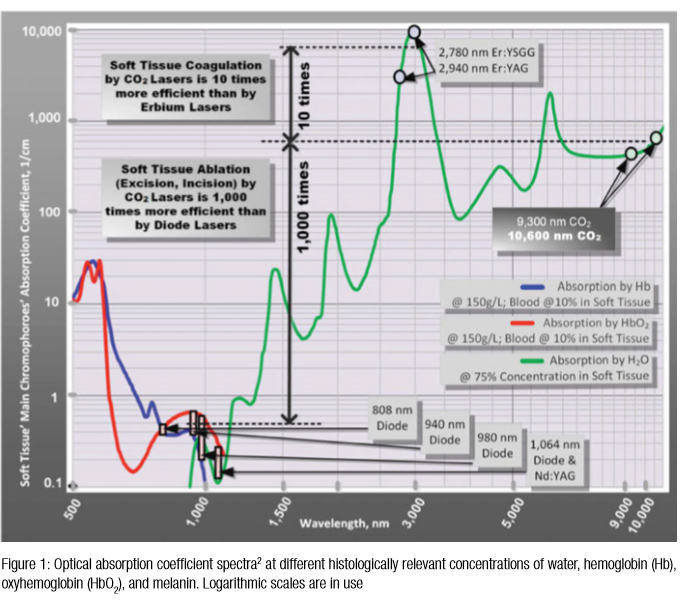 Soft tissue surgical lasers have many advantages over traditional scalpel surgery, cryosurgery, and electrosurgery. However, not all lasers are equally efficient at both cutting the soft tissue, coagulating, and hemostasis because light absorption in the soft tissue varies with wavelength.1-3 As illustrated in Figure 1, some dental laser wavelengths (around 1,000 nm, such as diodes and Nd:YAG) are efficient coagulators, but inefficient scalpels since they are poorly absorbed by the soft tissue. Other dental laser wavelengths (around 3,000 nm, such as Erbium lasers) are well absorbed by the water-rich soft tissue and are great at cutting, but are not as efficient at coagulating and hemostasis.
Soft tissue surgical lasers have many advantages over traditional scalpel surgery, cryosurgery, and electrosurgery. However, not all lasers are equally efficient at both cutting the soft tissue, coagulating, and hemostasis because light absorption in the soft tissue varies with wavelength.1-3 As illustrated in Figure 1, some dental laser wavelengths (around 1,000 nm, such as diodes and Nd:YAG) are efficient coagulators, but inefficient scalpels since they are poorly absorbed by the soft tissue. Other dental laser wavelengths (around 3,000 nm, such as Erbium lasers) are well absorbed by the water-rich soft tissue and are great at cutting, but are not as efficient at coagulating and hemostasis.
Why CO2 laser?
The 10,600 nm CO2 laser wavelength it is not as good as Erbium laser at cutting (but is a much better coagulator2) and not as good as diode/Nd:YAG laser wavelengths at coagulating (but is a much better scalpel2,3). As indicated in Figure 1, the 10,600 nm CO2 laser wavelength is a compromise for simultaneous cutting and coagulation/hemostasis; most importantly, the 10,600 nm CO2 laser’s coagulation depth closely matches blood capillary diameters2. The radiant energy of the CO2 laser is used directly to photo-thermally vaporize (cut, incise, ablate) and, at the same time, to photo-thermally coagulate the soft tissues. The CO2 laser is a “what-you-see-is-what-you-get” surgical soft tissue laser with minimal collateral thermal effects that are sufficient for sealing blood vessels, lymphatics, and nerve endings; the surface bacteria are efficiently destroyed4 on incision/ablation margins.
Laser handpieces and ergonomics
The CO2 surgical lasers in the 1970s-1980s utilized the articulated arm beam delivery systems. The flexible, hollow fibers, introduced in the 1990s, significantly simplified CO2 laser surgery. Modern flexible fiber CO2 lasers use a scalpel-like ergonomic autoclavable handpiece (Figures 2 and 3) for 1) cutting, 2) ablation, and 3) photo-thermal coagulation and hemostasis. Tipless CO2 handpieces are designed to operate without disposables. The right-angle, pen-sized tipless handpiece permits easy and convenient access to the soft tissue within the oral cavity in patients of all ages, including infants.5
Photo-thermal ablation and coagulation
The photo-thermal ablation of soft tissue is a process of vaporizing intra- and extra-cellular water near the surface, where the laser beam intensity is at its maximum. For a fixed laser beam diameter (or spot size), the volume of the tissue exposed to laser beam is proportional to the optical penetration depth (inverse of absorption coefficient from Figure 1). The 10,600 nm CO2 laser is energy efficient at ablating the soft tissue photo-thermally with low ablation threshold intensities due to very small volume of irradiated tissue (because of extremely short absorption depth around 15 µm).2 Immediately below the ablation zone, the coagulation zone is located where the denaturation of soft tissue proteins occurs in 60°C–100°C temperature range. This process leads to a significant reduction in bleeding and oozing of lymphatic liquids on the margins of ablated tissue. For the CO2 laser, its excellent coagulation efficiency is due to the close match between the sub-100 µm6 photo-thermal coagulation depth2 and the oral soft tissue blood capillary diameters of approximately 20-40 µm7.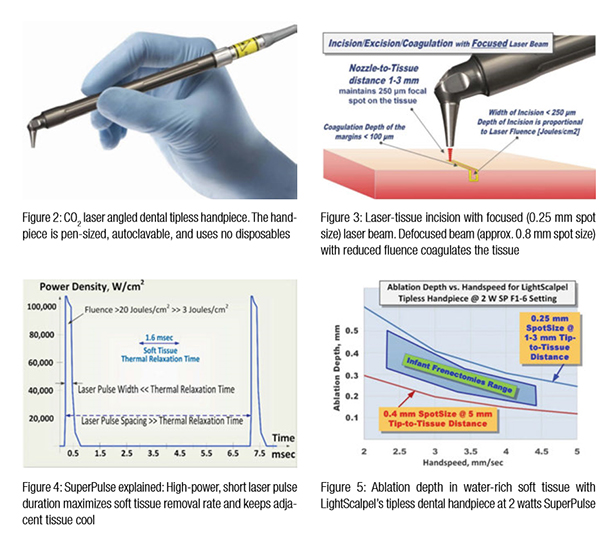
The many uses for a soft tissue CO2 laser in orthodontics, such as gingivectomies, frenectomies, exposure of impacted teeth, and others, feature minimal blood loss and reduced discomfort for the patients.
Laser pulsing and controlling thermal effects
Laser pulsing is as important for laser surgery as the wavelength — short and powerful pulses are often superior to long and weak ones. The exact physics of pulsed laser surgery deals with the Thermal Relaxation Time,2,3 which depends both on tissue’s light absorption and thermal diffusivity, first described by Einstein.8 The rate at which the irradiated tissue diffuses the heat away is defined by Thermal Relaxation Time TR, which equals approximately 1.5 msec for 75% water-rich soft tissue irradiated by the 10,600 nm CO2 laser. Practical implications of the Thermal Relaxation Time concept are simple, yet crucial for the appropriate application of laser energy. The irradiated tissue heats most efficiently when the energy of a laser pulse is high, and the duration of a pulse is much shorter than TR. The tissue adjacent to the ablated zone cools down most efficiently when time duration between laser pulses significantly exceeds TR. Such laser pulsing is referred to as SuperPulse (see Figure 4). SuperPulse minimizes the depth of coagulation and is therefore a must-have feature of any state-of-the-art soft tissue surgical CO2 laser. The optimal combination of the CO2 laser wavelength and pulsing results in a char-free and bloodless surgery. This also allows for a scar-free, uncomplicated healing that is valued by surgeons across all specialties such as dentistry, OMFS, ENT, and plastic surgery.
Laser beam spot size for cutting and coagulation
Just like the sharpness of the steel blade defines the quality and ease of the incision, the size of the laser beam focal spot defines the quality of the laser cut. The smaller (or sharper) the focal spot of the beam, the narrower and deeper the incision. For cutting, the LightScalpel laser handpiece is maintained 1 mm–3 mm away from the tissue and is moved at a hand speed of a few millimeters per second as illustrated in Figure 3. Just like a dull blade cannot produce a quality incision, an oversized laser beam spot cannot produce a precise and narrow cut. For a rapid switch from cutting to photo-coagulation alone, the laser beam can be de-focused either by selecting a larger spot size, or by simply moving the handpiece away from the tissue by approximately 10 mm (for LightScalpel tipless laser handpieces), and “painting” the “bleeder” for enhanced hemostasis.
Laser power density and depth of ablation
For a laser scalpel, the power density of the focused laser beam is equivalent to the mechanical pressure that is applied to a cold steel blade. In other words, greater laser fluence3 (i.e., higher power density and slower hand speed) results in greater depth and rate of soft tissue removal. During each SuperPulse pulse, the ablation depth δ is given by the formula δ = A (E – Eth) / Eth for the steady state ablation conditions3 where A is the absorption depth from Figure 1 and Eth is the ablation threshold fluence,3 and E is the fluence during the SuperPulse pulse. At the 10.6 µm wavelength of the CO2 laser, the ablation threshold for a water-rich soft tissue with an assumed water content of 75% equals approximately Eth = 3 J/cm2. For repetitive pulses that are scanned across the soft tissue, the fluence is defined by the pulse frequency and the hand speed: i.e., the depth of incision depends on laser power settings, spot size, and the surgeon’s hand speed (Figure 5).
Two important built-in safety features are 1) sub-millimeter superficial depth of ablation and 2) approximately 0.2 mm ablation depth variation as distance to tissue changes in 1 mm–5 mm range, which makes the CO2 laser a gentler and more forgiving tissue vaporization instrument in comparison with a scalpel, electrosurge, or diode surgery. Also significant is the switch from ablation to coagulation mode by defocusing the beam, with laser beam fluence dropping below the ablation threshold of 3 Joules/cm2.2
CO2 laser use in the presence of orthodontia hardware
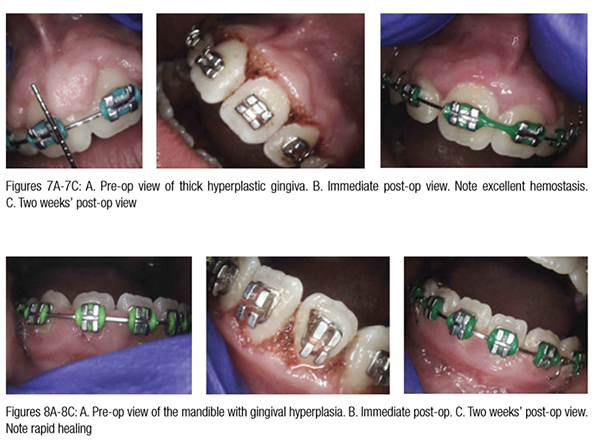
Gingival hypertrophy (caused by ortho-dontic positioning and poor oral hygiene) as well as overlying operculum are common problems during orthodontic therapy.9-11 Drug-induced hyperplasia presents another challenge for some orthodontic patients.13 Additionally, orthodontic devices attract bacteria and thus exacerbate the inflammation; periodontal disease often worsens in adolescents with fixed orthodontic hardware.9-13 Figure 6 demonstrates completely bloodless CO2 laser gingivectomy around the mandibular incisors in the orthodontic patient. CO2 laser gingivectomies, such as the ones presented in Figures 6-8, help improve the esthetics and oral health; they also allow for more ideal bracket placement and quicker treatment with fewer appointments.


 Gaining access to an impacted tooth or teeth is often necessary in order to place orthodontic braces. With conventional scalpel surgery, many problems occur when placing brackets, such as sutures breaking before brackets are bonded. Brackets will not bond properly if enamel is wet due to bleeding. Clean, bloodless enamel is necessary for enamel acid etching and bonding of the composite resin. Using the CO2 laser to expose an impacted tooth produces a dry surgical field thus creating conditions required for immediate bracket bonding. The CO2 laser-assisted cuspid exposure procedure is shown in Figure 9. This quick CO2 laser treatment allows access for bracket placement, saving the patient months of waiting for the tooth to erupt on its own. The CO2 laser provides a clean, non-bleeding border. The CO2 laser straight and angled handpieces, illustrated in Figures 2 and 3, permit a convenient reach into areas that may be obstructed by braces. Once an impacted tooth is exposed by laser ablation, the bloodless surgical field is ready for the next step. The orthodontic traction hook can be bonded to the exposed enamel immediately after ablation during the same visit,10,11 saving time for both the patient and clinician.
Gaining access to an impacted tooth or teeth is often necessary in order to place orthodontic braces. With conventional scalpel surgery, many problems occur when placing brackets, such as sutures breaking before brackets are bonded. Brackets will not bond properly if enamel is wet due to bleeding. Clean, bloodless enamel is necessary for enamel acid etching and bonding of the composite resin. Using the CO2 laser to expose an impacted tooth produces a dry surgical field thus creating conditions required for immediate bracket bonding. The CO2 laser-assisted cuspid exposure procedure is shown in Figure 9. This quick CO2 laser treatment allows access for bracket placement, saving the patient months of waiting for the tooth to erupt on its own. The CO2 laser provides a clean, non-bleeding border. The CO2 laser straight and angled handpieces, illustrated in Figures 2 and 3, permit a convenient reach into areas that may be obstructed by braces. Once an impacted tooth is exposed by laser ablation, the bloodless surgical field is ready for the next step. The orthodontic traction hook can be bonded to the exposed enamel immediately after ablation during the same visit,10,11 saving time for both the patient and clinician.
Many patients require banding of molars, but an overlying operculum gets in the way of placing appliances on these teeth. The CO2 laser is an excellent tool to remove this excess tissue. Hemostasis allows for banding to happen immediately following CO2 treatment. Local anesthetic may be required in some cases. Figure 15 presents theLightScalpel LS-1005 CO2 laser operculectomy (3 watts SuperPulse Repeat Mode F1-7) prior to band placement. Bands were seated shortly after operculectomy was performed, in the same visit.
An important safety consideration is the heating rate of the orthodontia that could be accidentally exposed to a direct laser beam. Due to the differences in the light reflectance of stainless steel at different wavelengths,14 the wavelengths of approximately 800 nm–3,000 nm (diode, Nd:YAG and Erbium lasers) are absorbed 50%-250% stronger than CO2 laser wavelengths around 10,000 nm. Such high reflectivity by stainless steel makes the CO2 laser the safest wavelength around stainless steel orthodontia. When compared to lasers, electrosurgical units present a different safety challenge of conducting electrical currents by orthodontic hardware during accidental contacts with electro-surgical electrodes.
Minimized bleeding for enhanced visibility and precision
The enhanced hemostatic ability and precision of the CO2 laser is especially valuable in visible (esthetic) zones. The CO2 laser surgical removal of the tissue presented in Figures 6-15 illustrates excellent coagulation and hemostasis due to a close match between the oral soft tissue blood capillaries’ diameters of 20-40 µm7 and the coagulation depth during soft tissue ablation.2 Incisions with minimal bleeding, or entirely bloodless incisions, provide great visibility of the cut site, and thus higher precision for a minimized tissue trauma and better esthetic outcome.9-13,15-17
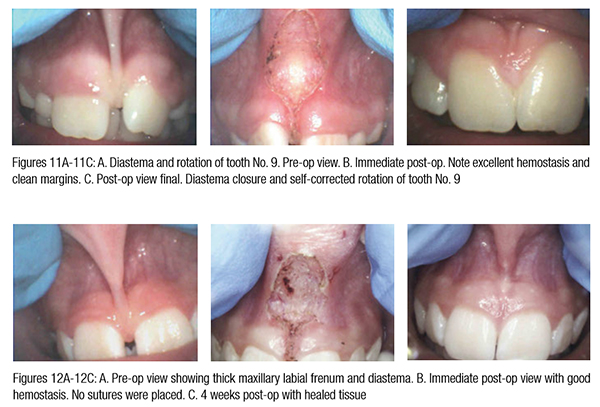
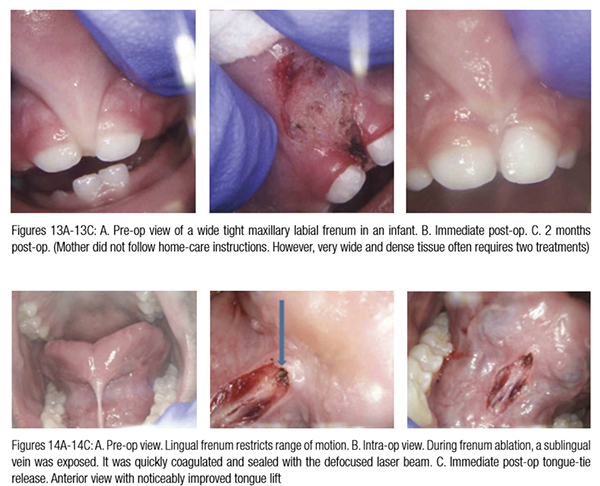
For example, Figure 10 shows the use of LightScalpel LS-1005 CO2 laser (2 watts SuperPulse Repeat Mode with F1-6 through F1-8 settings) for soft tissue crown lengthening of the maxillary incisors and for labial frenum revision. Both procedures were performed during the same visit while the patient was under anesthesia. No sutures were placed. Figure 11 presents the self-correction of a rotated tooth No. 9 after frenum revision. Laser frenectomies in Figures 12 and 13 resulted in spontaneous closure of midline diastemas.
In comparison with traditional scalpel surgery, the precision of CO2 laser ablation allows for sub-millimeter control over tissue vaporization depth (Figure 5). The CO2 laser accuracy is especially important for highly vascularized areas. For example, the 14-year-old patient in Figure 14 had a CO2 laser lingual frenectomy performed during which a sublingual vein was exposed and coagulated (sealed) with a defocused laser beam. As a result, no sutures were needed, and the surgical wound was left to heal by secondary intention.
Tissue healing
With CO2 laser-assisted oral surgery, sutures are often not necessary, unlike with scalpel procedures. Laser wounds are often left to heal by secondary intention, as presented in Figures 6-14. Studies have shown that there is a diminished risk of scarring and wound contraction associated with CO2 laser surgery.18-22 Laser-induced wounds heal with greater fibroblast proliferation, with young fibroblasts actively producing collagen. Laser wounds also have been reported to contain smaller amounts of myofibroblasts (cells responsible for wound contraction), compared to scalpel wounds.22-24 Secondary intention healing and lack of scarring are especially important for treating lesions located in esthetic/visible zones.
Reduced postoperative pain and discomfort
It is important to avoid charring or causing excessive thermal damage to soft tissue. The experienced CO2 laser surgeon using the proper laser settings and hand speed will have small (sub 50 microns thick) zones of thermal necrosis.5 Patients report less postoperative pain and discomfort with laser surgery.25 The healing process associated with CO2 laser surgery is enhanced and less painful than with cryosurgery or electrosurgery.20-21
Summary
The many uses for a soft tissue CO2 laser in orthodontics, such as gingivectomies, frenectomies, exposure of impacted teeth, and others, feature minimal blood loss and reduced discomfort for the patients. In comparison with other dental laser wavelengths (circa 1,000 nm for diode and Nd:YAG lasers and circa 3,000 nm for Erbium lasers), the CO2 laser wavelength exhibits the least absorption rate by the stainless steel orthodontia hardware, and the optimum coagulation depth closely matching gingival blood vessel diameters. The CO2 laser ablation depth is controlled to a few tenths of a millimeter, which characterizes this wavelength as a safe and gentle soft tissue removal tool, while the sub-100 micrometer coagulation depth (better than diodes and electrosurgery) allows for enhanced, scar-free healing of highly vascular oral tissues (often by secondary intention). The accuracy and precision of CO2 laser surgery are increased by the visibility of the surgical field not being obscured by bleeding.
Acknowledgments
Authors greatly appreciate the help and contribution from Anna (Anya) Glazkova, PhD, and Olga Vitruk, BSc, in preparing this material for publication.
- Jacques SL. Optical properties of biological tissues: a review. Phys Med Biol. 2013;58(11):R37-61.
- Vitruk P. Oral Soft Tissue Laser Ablative & Coagulative Efficiencies Spectra. Implant Practice US. 2014;7(6):22-27.
- Vogel A, Venugopalan V. Mechanisms of pulsed laser ablation of biological tissues. Chem Rev. 2003;103(2):577-644.
- Cobb C, Vitruk P. Effectiveness of a Super Pulsed CO2 Laser for Removal of Biofilm from Three Different Types of Implant Surfaces: An In Vitro Study. Implant Practice US. 2015;8(3):20-28.
- Kaplan M, Hazelbaker A, Vitruk P. Infant Frenectomy with 10,600 nm Dental CO2 Laser. Washington Academy of General Dentistry newsletter. 2015;April(34):10-12.
- Wilder-Smith P, Arrastia AM, Liaw LH, Berns M. Incision properties and thermal effects of three CO2 lasers in soft tissue. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 1995;79(6):685-691.
- Yoshida S, Noguchi K, Imura K, Miwa Y, Sunohara M, Sato I. A morphological study of the blood vessels associated with periodontal probing depth in human gingival tissue.Okajimas Folia Anat Jpn. 2011;88(3):103-109.
- Einstein A. Über die von der molekularkinetischen theorie der wärme geforderte bewegung von in ruhenden flüssigkeiten suspendierten teilchen. Ann Phys.1905;322(8):549-560.
- Gama SK, de Araujo TM, Pinheiro AL. Benefits of the use of the CO2 laser in orthodontics. Lasers Med Sci. 2008;23(4):459-465.
- Convissar RA, Diamond LB, Fazekas CD. Laser treatment of orthodontically induced gingival hyperplasia. Gen Dent. 1996;44(1):47-51.
- Chmura LG. Soft tissue lasers in orthodontics. In: Convissar RA. Principles and Practice of Laser Dentistry. St. Louis, MO: Mosby; 2011:225-242.
- Monteiro LS, Mouzinho J, Azevedo A, Infante da Camara M, Martins MA, Fuente-Lanos J. Treatment of epulis fissuratum with carbon dioxide laser. Rev Port Estomatol Med Dent Cir Maxilofac. 2011;52(3):165-169.
- Convissar RA. The top ten myths about CO2 lasers in dentistry. Dent Today. 2009;28(4):68, 70, 72-76.
- Bergman TL, Dewitt DP, Incropera FP, Lavine AS. Radiation: Processes and Properties. Fundamentals of Heat and Mass Transfer. 7th ed. Hoboken, NJ: John Wiley & Sons; 2011:768-860.
- Deppe H, Horch HH. Current status of laser applications in oral and cranio-maxillofacial surgery. Med Laser Appl. 2007;22(1):39-42.
- Strauss RA, Fallon SD. Lasers in contemporary oral and maxillofacial surgery. Dent Clin North Am. 2004;48(4):861-888.
- Namour S. Atlas of Current Oral Laser Surgery. Boca Raton, FL:Universal Publishers. 2011;121-135.
- Zaffe D, Vitale MC, Martignone A, Scarpelli F, Botticelli AR. Morphological histochemical and immunocytochemical study of CO2 and Er:YAG laser effect on oral soft tissues. Photomed Laser Surg. 2004;22(3):185-189.
- Zeinoun T, Nammour S, Dourov N, Aftimos G, Luomanen M. Myofibroblasts in healing laser excision wounds. Lasers Surg Med. 2001;28(1):74-79.
- Mason C, Hopper C. The use of CO2 laser in the treatment of gingival fibromatosis: a case report. Int J Paediatr Dent. 1994;4(2):105-109.
- Wang X, Ishizaki NT, Matsumoto K. Healing process of skin after CO2 laser ablation at low radiance: a comparison of continuous-wave and pulsed mode. Photomed Laser Surg. 2005;23(1):20-26.
- Grbavac RA, Veeck EB, Bernard JP, Ramalho LM, Pinheiro AL. Effects of laser therapy in CO2 laser wounds in rats. Photomed Laser Surg. 2006;24(3):389-396.
- de Freitas AC, Pinheiro AL, de Oliveira MG, Ramalho LM. Assessment of the behavior of myofibroblasts on scalpel and CO2 laser wounds: an immunohistochemical study in rats. J Clin Laser Med Surg.2002;20(4):221-225.
- Fisher SE, Frame JW, Browne RM, Tranter RM. A comparative histological study of wound healing following CO2 laser and conventional surgical excision of canine buccal mucosa. Arch Oral Biol. 1983;28(4):287-291.
- Tuncer I, Ozçakir-Tomruk, Sencift K, Cöloğlu S. Comparison of conventional surgery and CO2 laser on intraoral soft tissue pathologies and evaluation of the collateral thermal damage. Photomed Laser Surg. 2010;28(1):75-79.
Stay Relevant With Orthodontic Practice US
Join our email list for CE courses and webinars, articles and mores

 Dr. Martin Kaplan, DMD, practices exclusively in Stoughton, Massachusetts. Dr. Kaplan completed his undergraduate degree at the University of Massachusetts and then attended Tufts University School of Dental Medicine. After earning his DMD degree, he completed a pediatric residency at Montefiore Hospital in New York. Dr. Kaplan is an Adjunct Clinical Instructor at Tufts University School of Dental Medicine in the Postgraduate Pediatric Dental and Orthodontic Departments. He is a member of the American Academy of Pediatric Dentistry, Massachusetts Academy of Pediatric Dentistry, American Dental Association, Academy of Laser Dentistry, Massachusetts Dental Society, Academy of Sports Dentistry, Massachusetts Breastfeeding Coalition, and Breastfeeding USA. Dr. Kaplan continues to take advanced training and recently gained masters’ proficiency in Laser Dentistry. He is one of only a handful of pediatric laser dentists. When not practicing dentistry, Dr. Kaplan enjoys karate and has attained the level of fourth-degree black belt.
Dr. Martin Kaplan, DMD, practices exclusively in Stoughton, Massachusetts. Dr. Kaplan completed his undergraduate degree at the University of Massachusetts and then attended Tufts University School of Dental Medicine. After earning his DMD degree, he completed a pediatric residency at Montefiore Hospital in New York. Dr. Kaplan is an Adjunct Clinical Instructor at Tufts University School of Dental Medicine in the Postgraduate Pediatric Dental and Orthodontic Departments. He is a member of the American Academy of Pediatric Dentistry, Massachusetts Academy of Pediatric Dentistry, American Dental Association, Academy of Laser Dentistry, Massachusetts Dental Society, Academy of Sports Dentistry, Massachusetts Breastfeeding Coalition, and Breastfeeding USA. Dr. Kaplan continues to take advanced training and recently gained masters’ proficiency in Laser Dentistry. He is one of only a handful of pediatric laser dentists. When not practicing dentistry, Dr. Kaplan enjoys karate and has attained the level of fourth-degree black belt. Peter Vitruk, PhD, MInstP, CPhys, is a founder of LightScalpel, LLC, in Woodinville, Washington. He is a member of The Institute of Physics, United Kingdom, and is the member of the Science and Research Committee, Academy of Laser Dentistry, United States. He is also on the faculty of Global Laser Oral Health, U.S. Dr. Vitruk can be reached at 1-866-589-2722 or pvitruk@lightscalpel.com.
Peter Vitruk, PhD, MInstP, CPhys, is a founder of LightScalpel, LLC, in Woodinville, Washington. He is a member of The Institute of Physics, United Kingdom, and is the member of the Science and Research Committee, Academy of Laser Dentistry, United States. He is also on the faculty of Global Laser Oral Health, U.S. Dr. Vitruk can be reached at 1-866-589-2722 or pvitruk@lightscalpel.com.
